Botox Cosmetic
Real results that are true to you
Are you ready to discover real, noticeable results? Learn more about BOTOX® Cosmetic (onabotulinumtoxinA) and decide if it’s time to ask your doctor about treatment.
What is BOTOX® Cosmetic?
BOTOX® Cosmetic is the first and only FDA-approved product to temporarily improve both moderate to severe frown lines between the brows and crow’s feet lines around the sides of the eyes in adults.
How does BOTOX® Cosmetic work?
BOTOX® Cosmetic targets one of the underlying causes of frown lines and crow’s feet—the repeated muscle contractions from frowning and squinting over the years. Your doctor will inject these muscles with BOTOX® Cosmetic to temporarily reduce muscle activity. You’ll begin to notice a visible smoothing of these lines.
Did YOU know?
Important facts about BOTOX® Cosmetic (onabotulinumtoxinA):
- In 2002, BOTOX® Cosmetic became the first product of its kind in the United States to receive FDA approval for moderate to severe frown lines
- In 2013, it became the first and only product to receive FDA approval for moderate to severe crow’s feet
- #1 selling product of its kind in the world (2),*
- ≈ 11 million treatments for moderate to severe frown lines have been performed since 2002 (3)
- Approved in 78 countries for aesthetic use (4),†
*Data collected through December 2013.
†VISTABEL® in most European countries.
People, just like you
- Approximately 8 out of 10 surveyed neurotoxin patients (n = 276) expect to receive BOTOX® Cosmetic (onabotulinumtoxinA) at their next treatment (5)
- Approximately 9 out of 10 BOTOX® Cosmetic surveyed patients (n = 300) are willing to tell friends and family about their treatment (5)
- Plus, BOTOX® Cosmetic has a record of experience that aesthetic providers know and trust. Is it time to ask your doctor about BOTOX® Cosmetic?

Senara is proud to participate in the Allergan Brilliant Distinctions program.
What to Expect with Treatment
How is BOTOX® Cosmetic (onabotulinumtoxinA) administered?
To temporarily improve moderate to severe crow’s feet, your doctor will inject BOTOX® Cosmetic into the muscle surrounding the sides of your eyes—called the orbicularis oculi.
For temporary improvement of moderate to severe frown lines, BOTOX® Cosmetic is injected into 2 muscles—the corrugator and procerus muscles.
Treatment with BOTOX® Cosmetic takes approximately 10 minutes and requires minimal downtime or recovery it’s often called a lunchtime procedure.
Think 2 treatments, 1 visit. Ask your doctor about receiving BOTOX® Cosmetic for moderate to severe frown lines and crow’s feet at the same time.1
Will BOTOX® Cosmetic make me look like I’ve had work done?
When BOTOX® Cosmetic is administered by an experienced injector, you should not lose the ability to show expressions. BOTOX® Cosmetic is a techniquesensitive procedure that should only be performed by a licensed and trained aesthetic provider who has expertise in facial anatomy.
Talk to your doctor about your expectations and the results you want to achieve with treatment.
Is BOTOX® Cosmetic (onabotulinumtoxinA) for me?
Take some time to read through this webpage, visit BotoxCosmetic.com, and talk to your doctor.
What are the most common side effects of BOTOX® Cosmetic?
The most common side effects are eyelid drooping or swelling. Other possible side effects include: dry mouth; discomfort or pain at the injection site; tiredness; headache; neck pain; eye problems: double vision, blurred vision, decreased eyesight, and dry eyes; and allergic reactions.(1)
Symptoms of an allergic reaction to BOTOX® Cosmetic may include: itching, rash, red itchy welts, wheezing, asthma symptoms, or dizziness or feeling faint. Tell your doctor or get medical help right away if you are wheezing or have asthma symptoms, or if you become dizzy or faint. (1)
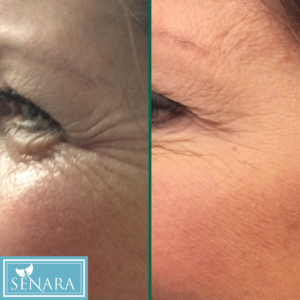
- See more real, noticeable results in the Before & After Gallery
- Watch real stories from people who have experienced BOTOX® Cosmetic (onabotulinumtoxinA)
BOTOX® Cosmetic (onabotulinumtoxinA) IMPORTANT SAFETY INFORMATION
Do not take BOTOX® Cosmetic if you: are allergic to any of the ingredients in BOTOX® Cosmetic (see Medication Guide for ingredients); had an allergic reaction to any other botulinum toxin product such as Myobloc® (rimabotulinumtoxinB), Dysport® (abobotulinumtoxinA), or Xeomin® (incobotulinumtoxinA); have a skin infection at the planned injection site.
1. BOTOX® Cosmetic Prescribing Information, September 2013. 2. Data on file, Allergan, Inc.,
December 2013. 3. Data on file, Allergan, Inc., March 25, 2009. 4. Data on file, Allergan, Inc.;
December 2013; BOTOX® and BOTOX® Cosmetic Country Approvals. 5. Facial Injectables
Consumer User Survey; May 2014. 6. Data on file, Allergan, Inc., December 30, 2013.
Actual patients. Results may vary.
Photos taken at full smile before and after treatment with BOTOX® Cosmetic (onabotulinumtoxinA) at day 7. In two clinical studies, 26.1% and 20.3% of people had ≥ 2-grade improvement at day 30.1 In one of these studies, 67.9% of people had mild or no crow’s feet at day 30 after treatment.2
Photos taken at maximum frown before and after treatment with BOTOX® Cosmetic (onabotulinumtoxinA) at day 7. In clinical studies, physicians assessed 80% of people had significant improvement at day 30.1 Photos taken at maximum frown and/or full smile before and after treatment with BOTOX® Cosmetic (onabotulinumtoxinA) at day 7. In two clinical studies for moderate to severe crow’s feet, 26.1% and 20.3% of people had ≥ 2-grade improvement at day 30.1 In one of these studies, 67.9% of people had mild or no crow’s feet at day 30 after treatment.2 In clinical studies for moderate to severe frown lines, physicians assessed 80% of people had significant improvement at day 30.1
Side effects associated with the injection include localized pain, infection, inflammation, tenderness, swelling, redness, and/or bleeding/bruising.1
BOTOX® Cosmetic is a prescription medicine that is injected into muscles and used to improve the look of moderate to severe frown lines between the eyebrows (glabellar lines) in adults for a short period of time (temporary).BOTOX® Cosmetic is a prescription medicine that is injected into the area around the side of the eyes to improve the look of moderate to severe crow’s feet lines in adults for a short period of time (temporary).
IMPORTANT SAFETY INFORMATION
BOTOX® Cosmetic may cause serious side effects that can be life threatening. Call your doctor or get medical help right away if you have any of these problems any time (hours to weeks) after injection of BOTOX® Cosmetic:
- Problems swallowing, speaking, or breathing, due to weakening of associated muscles, can be severe and result in loss of life. You are at the highest risk if these problems are pre-existing before injection. Swallowing problems may last for several months
- Spread of toxin effects. The effect of botulinum toxin may affect areas away from the injection site and cause serious symptoms including: loss of strength and all-over muscle weakness, double vision, blurred vision and drooping eyelids, hoarseness or change or loss of voice (dysphonia), trouble saying words clearly (dysarthria), loss of bladder control, trouble breathing, trouble swallowing. If this happens, do not drive a car, operate machinery, or do other dangerous activities
The dose of BOTOX® Cosmetic is not the same as, or comparable to, any other botulinum toxin product.
There has not been a confirmed serious case of spread of toxin effect when BOTOX® Cosmetic has been used at the recommended dose to treat frown lines, crow’s feet lines or both at the same time.
Serious and/or immediate allergic reactions have been reported. They include: itching, rash, red itchy welts, wheezing, asthma symptoms, or dizziness or feeling faint. Tell your doctor or get medical help right away if you are wheezing or have asthma symptoms, or if you become dizzy or faint.
Do not take BOTOX® Cosmetic if you: are allergic to any of the ingredients in BOTOX® Cosmetic (see Medication Guide for ingredients); had an allergic reaction to any other botulinum toxin product such as Myobloc® (rimabotulinumtoxinB), Dysport® (abobotulinumtoxinA), or Xeomin® (incobotulinumtoxinA); have a skin infection at the planned injection site.
Tell your doctor about all your muscle or nerve conditions, such as amyotrophic lateral sclerosis (ALS or Lou Gehrig’s disease), myasthenia gravis, or Lambert-Eaton syndrome, as you may be at increased risk of serious side effects including severe dysphagia (difficulty swallowing) and respiratory compromise (difficulty breathing) from typical doses of BOTOX® Cosmetic.
Tell your doctor about all your medical conditions, including: plans to have surgery; had surgery on your face; weakness of forehead muscles, such as trouble raising your eyebrows; drooping eyelids; any other abnormal facial change; are pregnant or plan to become pregnant (it is not known if BOTOX® Cosmetic can harm your unborn baby); are breast-feeding or plan to breast-feed (it is not known if BOTOX® Cosmetic passes into breast milk).
Tell your doctor about all the medicines you take, including prescription and nonprescription medicines, vitamins, and herbal products. Using BOTOX® Cosmetic with certain other medicines may cause serious side effects. Do not start any new medicines until you have told your doctor that you have received BOTOX® Cosmetic in the past.
Especially tell your doctor if you: have received any other botulinum toxin product in the last 4 months; have received injections of botulinum toxin, such as Myobloc®, Dysport®, or Xeomin® in the past (be sure your doctor knows exactly which product you received); have recently received an antibiotic by injection; take muscle relaxants; take an allergy or cold medicine; take a sleep medicine; take anti-platelets (aspirin-like products) or anti-coagulants (blood thinners).
Other side effects of BOTOX® Cosmetic include: dry mouth, discomfort or pain at the injection site, tiredness, headache, neck pain, and eye problems: double vision, blurred vision, decreased eyesight, drooping eyelids, swelling of your eyelids, and dry eyes.
For more information refer to the Medication Guide or talk with your doctor.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088.
Please see BOTOX® Cosmetic full Product Information including Boxed Warning and Medication Guide.
1. BOTOX® Cosmetic Prescribing Information, September 2013.
2. Data on file, Allergan, Inc., December 30, 2013.


